
CALCULATIONS I. Reconstituting Cytokines/Growth Factors Need To Supplement Cultures With Recombinant Growth factors/Cytokines Issues To Consider –Recombinant. - ppt download
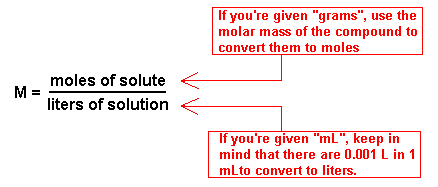
What is the molarity of a solution that contains 50.0 g of Mg(NO_3)_2 per 225 mL of solution? | Socratic

Question Video: Calculating the Molar Concentration of Mg(OH)₂ Using Data from a Titration Experiment | Nagwa
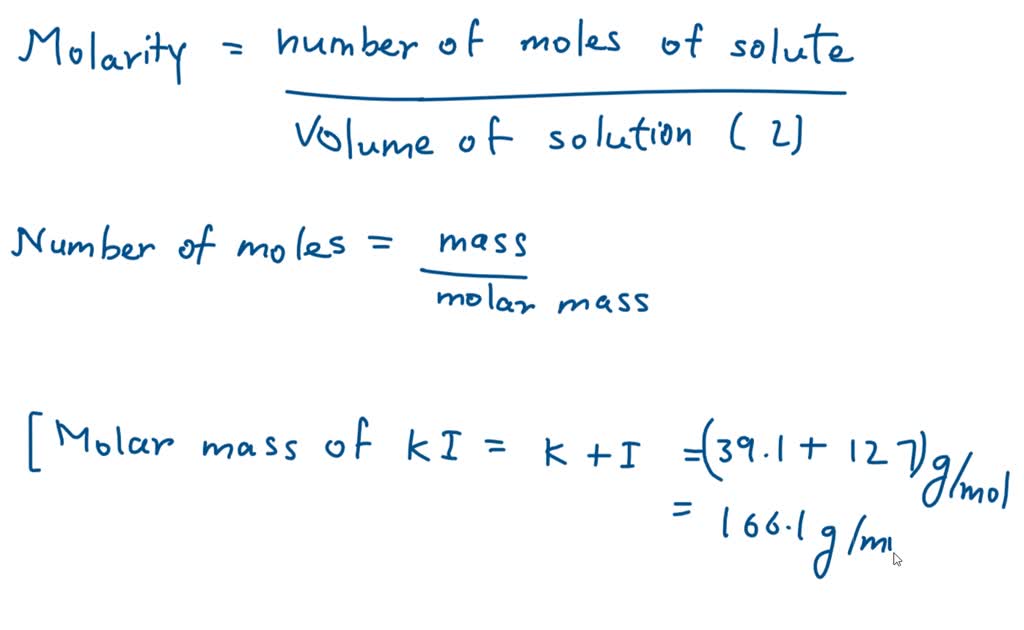
SOLVED: Calculate the molarity of each solution. 33.2 mg of KI in 118 mL of solution Express your answer using three significant figures.
![SOLVED: CHEM 121: Introduction to Chemistry Concentration is a conversion factor: Molarity [M, moles solute/L solution] lets you convert between volume of solution and moles of solute. Dilution Equation: M1V1 = M2V2. SOLVED: CHEM 121: Introduction to Chemistry Concentration is a conversion factor: Molarity [M, moles solute/L solution] lets you convert between volume of solution and moles of solute. Dilution Equation: M1V1 = M2V2.](https://cdn.numerade.com/ask_images/94726641ed1f4ed69c39f39139455c2d.jpg)
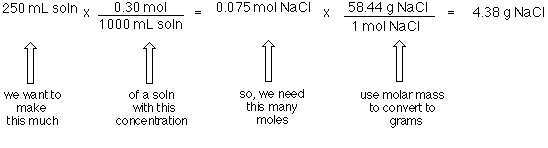



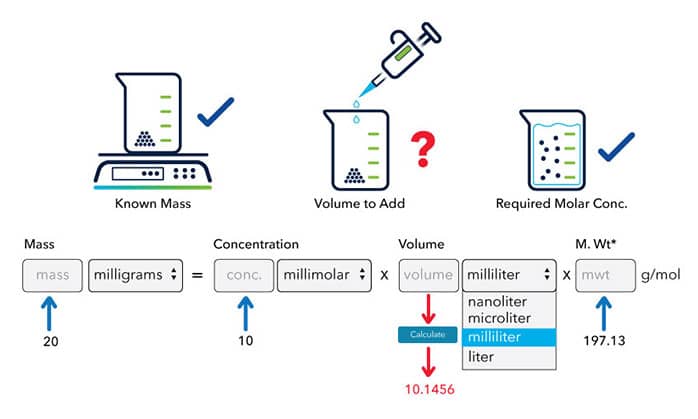

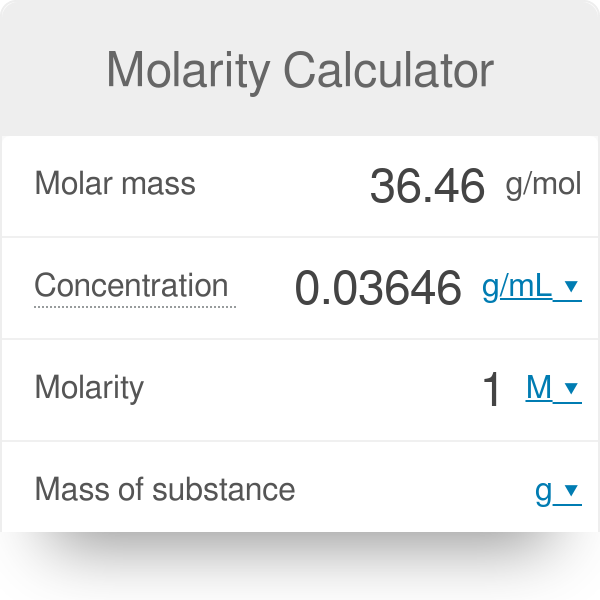
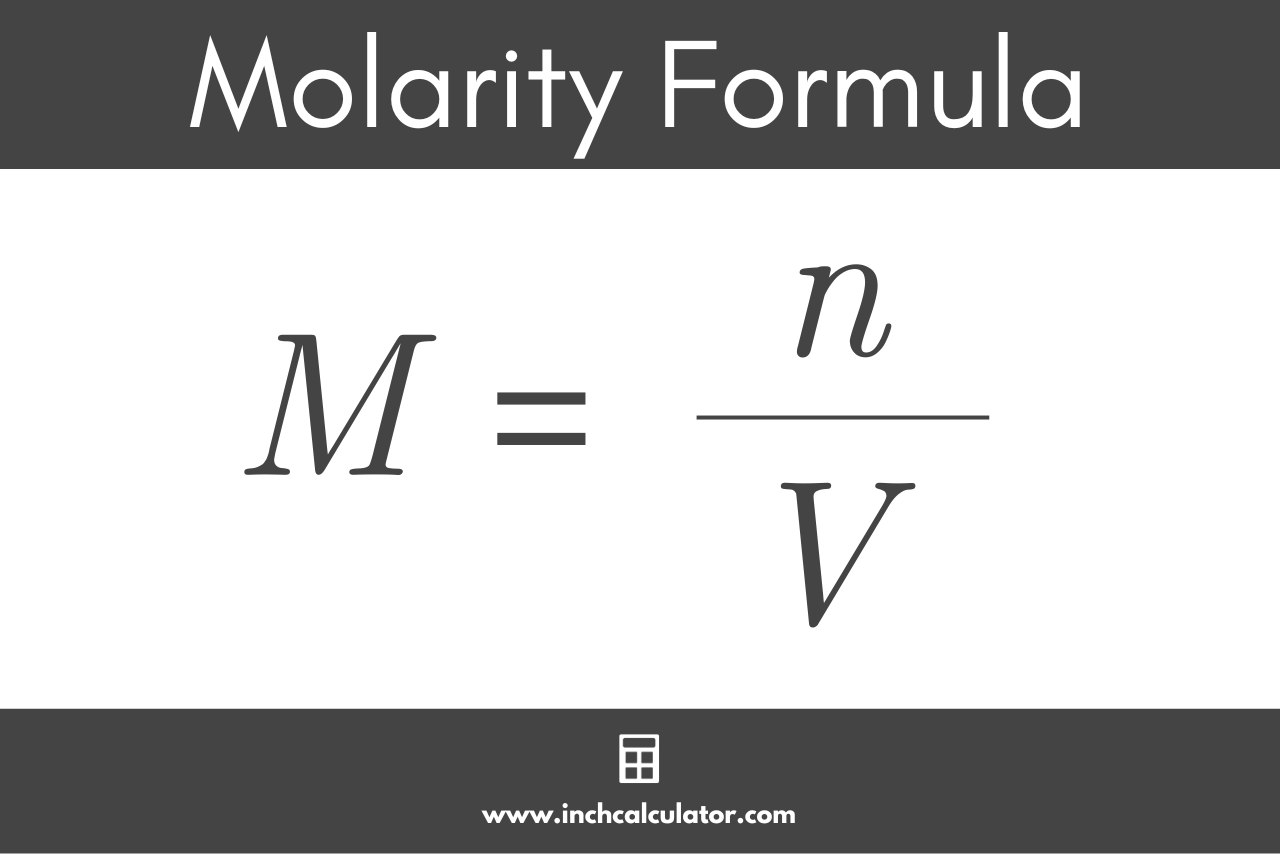





![Solved] To convert mg/mL to mM, use the formula ( | SolutionInn Solved] To convert mg/mL to mM, use the formula ( | SolutionInn](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2023/02/63e5fcb8cc19a_1676016823892.png)


